External Iliac Artery Endofibrosis
Background
External Iliac Artery Endofibrosis (EIAE) is a rare vascular condition that compromises blood flow to the lower limbs. It typically presents as thigh pain, cramping and weakness during high intensity exercise and settles shortly after cessation. Endofibrosis occurs when the intima (inner) layer of the artery becomes thickened and occludes blood flow. EIAE most commonly affects young healthy athletes, in particular cyclists and rowers- those who are least expected to experience vascular issues. This often results in delayed diagnosis or mis-management until symptoms progress and specialist opinion has been sought. It is thought a combination of repetitive hip flexion and high blood flows over thousands of hours of training causes flexing and microtrauma to the arterial wall, leading to endofibrotic changes.
Relevant Anatomy
The common iliac artery bifurcates at the level of the sacroiliac joint forming the left and right external iliac arteries. Each branch courses along the medial border of the psoas muscle towards the thigh, where they pass under the inguinal ligament and continue as the left and right femoral arteries. The external iliac arteries provide the main blood supply to the lower limb.
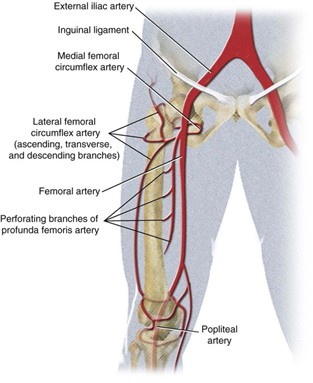
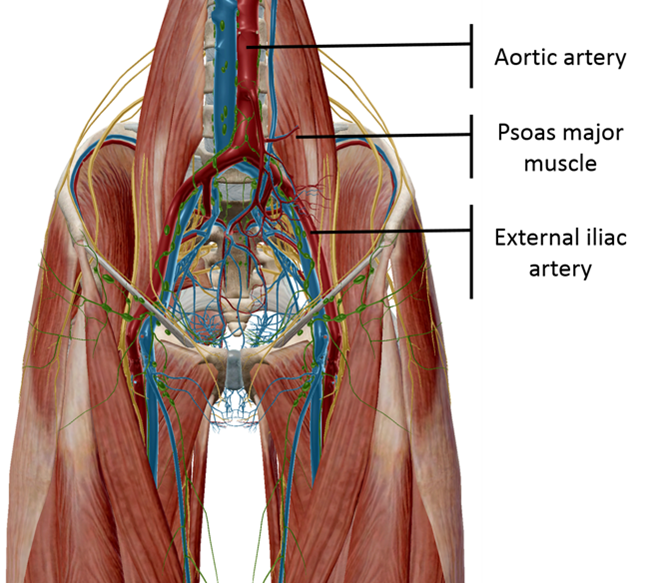
Image 1 and 2: Schematic diagrams of the EIA and surrounding anatomy
Causes
There are a number of factors linked to the development of EIAE:
Volume, intensity and type of training
Repetitive hip and trunk flexion movements associated with endurance sports such cycling and rowing cause direct mechanical stress on the EIA1. Aggressive aerodynamic positions requiring increased hip flexion angles (e.g. time trial position on the bike) further amplifies this effect. High internal arterial pressures and blood flows during intense efforts cause haemodynamic stress to the inner walls of the artery, further leading to endofibrotic changes2. Therefore, it is of no surprise that EIAE is most commonly seen in high level cyclists. Studies have shown that it is correlated with cycling training distance and intensity3.


Images 3 and 4: Endurance rowing and cycling place repetitive haemodynamic and mechanical stress on the EIA’s.
Psoas related factors
Due to its proximity to the EIA, psoas muscle hypertrophy can kink and stretch the artery during hip flexion3. Research has suggested that competitive cycling training can induce psoas muscle hypertrophy4. Similarly, arterial fixation to the psoas muscle or inguinal ligament can cause further arterial compression and mechanical stress.
Longer external iliac artery length
Longer artery length1 makes it kinking or folding of the artery more likely to occur during hip flexion. This is a non-modifiable risk factor that can occlude blood flow to the lower limbs, even in the absence of endofibrotic changes.
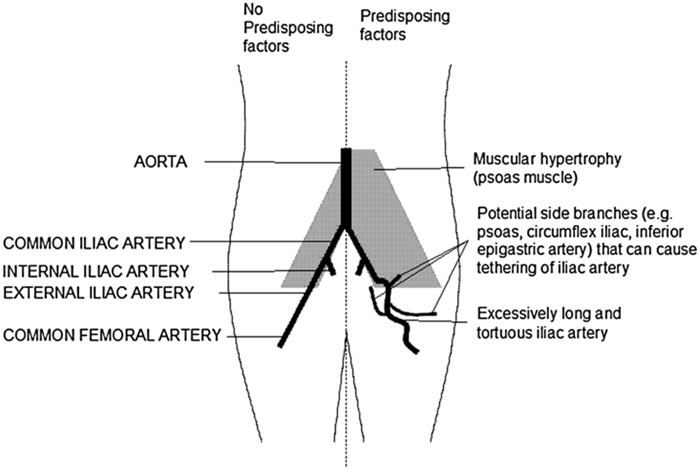
Image 5: Anatomical factors contributing to EIAE
Presentation
EIAE is most commonly seen in males1, which may be a reflection of their greater representation in high level cycling. It usually affects only one side, with the L) side being implicated in the majority of cases6.
Athletes are often asymptomatic at rest or submaximal exertion, with symptoms appearing at near maximal exertion. Thigh pain and leg weakness that resolves within 5 minutes of stopping exercise are the hallmark features of EIAE. Other commonly reported symptoms include cramping or paraesthesia in the thigh. In the early stages, symptoms only manifest during maximal or near maximal exertion (such as hill climbing, hard efforts or breakaways during racing) and settles with a relative drop in intensity. As the disease progresses, symptoms can be felt at lower intensities or by adopting positions of deeper hip flexion.
Diagnosis
A Delphi expert consensus study7 involving specialists with experience managing iliac endofibrosis (vascular surgeons and scientists, imaging specialists, sports scientists and physicians) came to agreement that:
- Leg weakness,
- Thigh pain,
- Resolution of symptoms within 5 minutes of cessation of exercise, are key diagnostic clinical findings of EIAE.
The preferred first line diagnostic investigation is doppler ultrasound. This non-invasive, accessible and risk-free modality uses high frequency sound waves to visualise blood flows through the EIA. This should be assessed before and immediate after exercise, ideally in hip flexion. Peak systolic velocities greater than 350cm/seconds post exercise are suggestive of severe vessel occlusion or stenosis.
Measuring Ankle Brachial Pressure Index (ABPI) before and after an exercise stress test is also a useful diagnostic tool to evaluate peripheral arterial perfusion to the lower limbs. The ABPI is calculated by dividing the highest systolic blood pressure at the ankle by the highest systolic pressure at the arm. Athletes with EAIE will generally have normal measurements (1.0-1.2) at rest, however, after exercise ABPI will drop as a result of reduced blood flow to the legs. It is important that exercise is continued until symptom reproduction, ABPI is measured within 1 minute of exercise cessation and in the supine position. Athletes in the early stages of EIAE will have to ride harder or longer during testing and may only experience a small drop in ankle pressures. In comparison, athletes with progressed EIAE may only have to ride for a few minutes at lower intensities to reproduce symptoms and experience greater pressure drops.
Other clinical signs/tests that can be used in conjunction with the above diagnostic tools and subjective presentation include:
- Weak or absent distal lower limb pulses after exercise.
- A bruit (abnormal sound heard under the stethoscope indicating partial obstruction of the blood vessel) heard over the femoral artery when the hip is held into flexion.
- CT or MR imaging with the hip in flexion to visually assess EIA length and kinking.
Management
Unfortunately, there are no conservative management strategies that can prevent or reverse EIAE pathology. Management will vary on a patient-by-patient basis- guided by goals, symptom severity and contributory factors. A Delphi consensus study7 concluded that cessation of symptom provoking exercise prevents disease progression, however, cannot reverse arterial pathoanatomical changes. Continuing to ride through symptoms will only accelerate endofibrotic changes. Therefore, conversation must be made between the clinician and patient regarding the cessation of high intensity/duration cycling.
In cases of early-stage disease, cyclists can prevent or delay the onset of symptoms by avoiding maximal intensity efforts (including climbing or racing) and minimising time spent in aggressive time trial positions. Optimising bike fit to ‘open up the hips’ can be achieved by increasing handlebar height or bringing the seat forward to reduce reach. This may come at an aerodynamic disadvantage for competitive cyclists. In the later stages of EIAE, cyclists may be only able to ride at lower intensities and in less challenging conditions.
More invasive interventions should be considered if clear arterial pathology is confirmed with imaging, symptoms negatively impact quality of life and the cyclist wishes to continue riding in the long term. Surgical consideration should be prefaced with the potential risks of such procedures involving major blood vessels (e.g. infection, arterial rupture, massive blood loss).
Surgical management will largely depend on the extent of endofibrotic thickening, contributing anatomical factors (including vessel length, psoas involvement) and goals of the athlete.
Expert consensus7 recommends open endarterectomy over endovascular approaches for most cases of EIAE. Endarterectomy involves excising the portion of affected arterial tissue then ‘patching up’ the opening with a vein harvested elsewhere in your body (usually the saphenous vein) or a synthetic patch. This procedure has been shown to have good outcomes with athletes being able to return to high level sports after a period of carefully monitored post operative management. There is a lack of research into long-term post operative outcomes.
Other less commonly used procedures include:
- bypass surgery to redirect blood flow around the affected blood vessel to the working lower limbs
- surgical release of the artery from the inguinal ligament or psoas muscle.
Post operative management
Depending on the extent of surgery, cyclists should restrain from usual training for at least 6-8 weeks and be guided by their surgeon. Rupture and subsequent internal haemorrhaging of the EIA can be catastrophic due to high blood flows. Similarly, infection after open surgery is a serious problem that requires urgent medical attention. Therefore, a period of rest and very gradual return to sport under close medical supervision is critical in the weeks following surgery. Cyclists should progress from walking to spinning on the trainer to easy road riding under close monitoring and medical supervision.
Summary
EIAE is a rare vascular condition that manifests as exercise induced thigh pain, weakness and cramping, with resolution of symptoms shortly after exercise cessation. It is characterised by thickening of the inner layer of the arterial wall of the external iliac artery- which supplies blood to the lower limbs. Repetitive hip flexion combined with high blood flows during high intensity exercise such as cycling causes mechanical stress to the EIA and is thought to accelerate endofibrotic changes.
A clinical diagnosis can be made with exercise induced symptoms and resolution within 5 minutes of cessation. Further investigations such as doppler ultrasound and ankle brachial pressure index can be used to confirm occlusion.
Conservative approaches aim to delay or prevent the onset of symptoms rather than addressing the actual pathology. These include intensity management and positional changes to minimise mechanical stress on the EIA.
Surgical management has been shown to be very effective, with high return to sport and competition rates, even at the highest level. Open endarterectomy with autologous patching is the preferred intervention, however, as with all major vascular surgery, presents with risks.

Julian Tubman (APAM)
Physiotherapist
Featured in the Top 50 Physical Therapy Blog
References
- Lim, C.S., Gohel, M.S., Shepherd, A.C., Davies, A.H. (2009) Iliac Artery Compression in Cyclists: Mechanisms, Diagnosis and Treatment. Eur J Endovasc Surg 38; 180-186
- Bender M,H., Schep, G., de Vries, W.R., Hoogeveen, A.R., Wijn, P.F. (2004) Sports-related flow limitations in the iliac arteries in endurance athletes: aetiology, diagnosis, treatment and future developments. Sports Med 34(7):427-42.
- Peach, G., Schep, G., Palfreeman, R., et al. (2012) Endofibrosis and kinking of the iliac arteries in athletes: A systematic review. Eur J Vasc Endovasc Surg; 43: 208–217.
- Ema, R., Wakahara, T., Yanaka, T., Kanehisa, H., Kawakami, Y.. (2016) Unique muscularity in cyclists’ thigh and trunk: A cross-sectional and longitudinal study. Scand J Med Sci Sports. 26(7):782-93
- Peach, G., Hinchliffe, R.J. Iliac Artery Endofibrosis in high performance athletes. Aspetar Sports Medicine Journal; 520-252
- Martinez, A., Wells, B.J. (2022) Vascular Disease Patient Information Page: External iliac artery endofibrosis. Vasc Med. 27(2): 207-210
- INSITE Collaborators (INternational Study group for Identification and Treatment of Endofibrosis).(2016) Diagnosis and Management of Iliac Artery Endofibrosis: Results of a Delphi Consensus Study. Eur J Vasc Endovasc Surg 52(1):90-8.
Image 1: https://radiologykey.com/vascular-anatomy-of-the-pelvis/
Image 2: https://posturemovementpain.com/2015/07/09/the-danger-of-the-drops-a-review-of-peach-et-al-2011/
Image 3: https://www.row2k.com/features/520/ease-into-the-catch/
Image 4: https://www.cyclingweekly.com/news/how-do-tour-de-france-time-trials-work
Image 5: https://www.ejves.com/article/S1078-5884(09)00180-4/fulltext#articleInformation







