Axial Spondyloarthritis – Diagnosis & Management
Definition
Axial spondyloarthritis is a chronic inflammatory rheumatic disease that affects the spine and sacroiliac joints. Axial spondyloarthritis is an umbrella term that covers two main types – ankylosing spondylitis and non-radiographic spondyloarthritis (1). Patients that have obvious structural changes on radiographs of the lumbar spine and sacroiliac joints are classified as radiographic axial spondyloarthritis or ‘Ankylosing Spondylitis’. Patients who are diagnosed with axial spondyloarthritis based on their symptoms and other clinical features, without radiographic structural changes are classified as non-radiographic axial spondyloarthritis. A small proportion of patients with non-radiographic axial spondyloarthritis will progress to AS over 5 years (4). Because AS is historically a more commonly recognised condition, much of the axial spondyloarthritis literature focuses on AS, rather than the less commonly recognised non-radiographic spondyloarthritis.
Diagnosis
Early diagnosis of axial spondyloarthritis remains a challenge, which leads to an average delay of 8.5 years to diagnose (1). Delays are often longer in women than men, as women tend to present with less “classical” symptomatology. The Assessment of SpondyloArthritis International Society (ASAS) published a set of criteria to aid in the diagnosis of axial spondyloarthritis as seen in the image below:
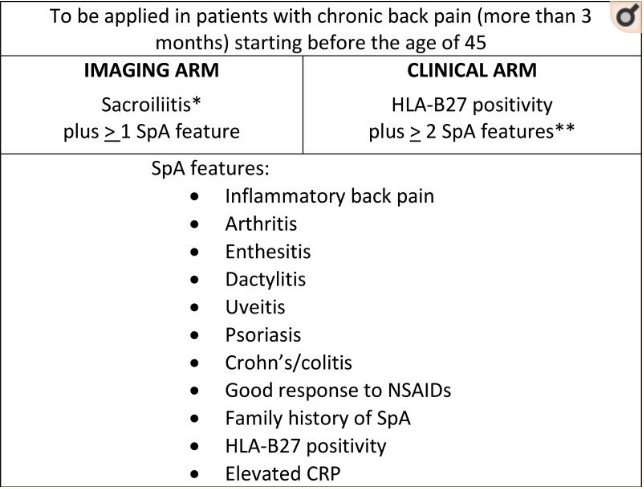
Clinical presentation
- Onset <40-45 years of age (peak incidence between 20-30 years)
- Back pain for >3 months
- Early morning spinal stiffness >30 minutes
- Improved pain with activity, worsened pain with rest
- Night pain in the 2nd half of the night
- Good response to non-steroidal anti-inflammatory medications (NSAID’s)
The above symptoms are indicative of inflammatory back pain, and require further specialist investigation by a rheumatologist.
Extra-articular features
- Acute anterior uveitis (inflammation of the iris) 30-40%
- Psoriasis 10%
- Inflammatory bowel disease 5-10%
- Osteoporosis in the spine peripheral joints leading to a fracture rate of 10%
Imaging
Chronic inflammation leads to lower back and sacroiliac joint pain and stiffness, which can lead to pathological new bone formation and ultimately fusion of the lumbar spine vertebrae and sacroiliac joints. A plain film x-ray of the lumbar spine and pelvis remains the best investigation to detect AS, as it is more sensitive to new bony growth than MRI. On x-ray imaging structural changes such as squaring of the vertebral bodies and bridging bone formation give the spine a bamboo-like appearance (1).
Ankylosing Spondylitis:
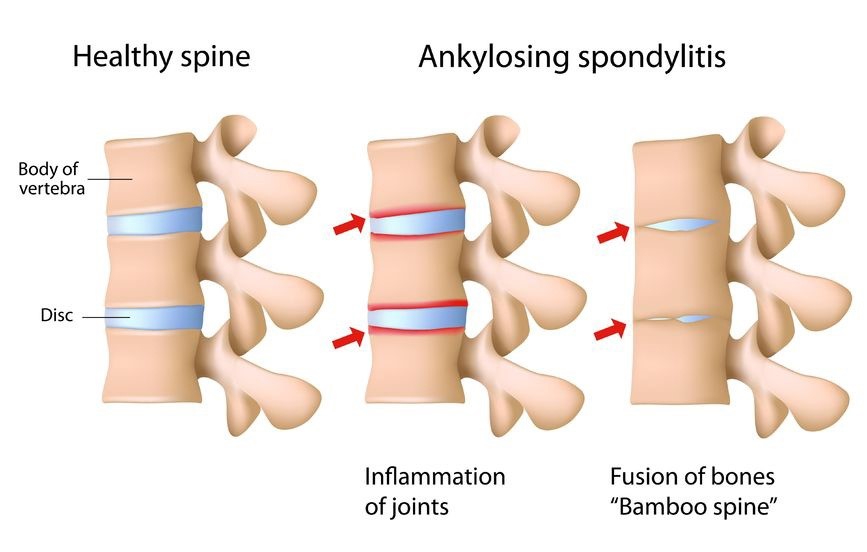
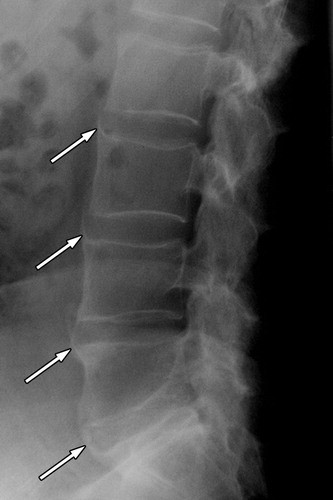
Although plain film X-ray remains the most sensitive radiograph to detect AS, MRI can be useful as it demonstrates inflammation in the early stages, which may or may not progress to bony structural changes visible on X-ray. Typical MRI findings include bone marrow oedema, fatty lesions, sclerosis or ankylosis on T1-weighted images. However, these findings alone are not necessarily indicative of non-radiographic axial spondyloarthritis, images need to be interpreted in the context of the entire clinical picture to make a diagnosis (1).
Genetic and Blood Markers
Axial spondyloarthritis is a genetically inherited disease, and twin studies have concluded that environmental risk factors play little role in the later development of axial spondyloarthritis (4, 5). Over 41 genes have currently been implicated in AS risk, however it is estimated that only about 25% of the genetic contributions to disease susceptibility is currently known (10). HLA-B27 is a class I surface antigen, which is found in 74 to 89% of patients with either non-radiographic axial spondyloarthritis or ankylosing spondylitis (8). However, HLA-B27 positivity is not sufficient for the diagnosis of AS, the risk of AS in populations that are HLA-B27 positive is between 2-10% (this increases if a first-degree relative is affected). Blood markers are used to assist diagnosis and monitor disease progression as well as treatment responses. C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) are the most commonly used blood markers in clinical practice and are elevated in approximately 40-50% of AS patients. Once again, these blood markers alone are not necessarily indicative of AS, the entire clinical picture must be considered (10).
Management
The overall goals of managing patients with AS is to maximise long-term health outcomes and quality of life. Treatment requires a multidisciplinary team approach under the care of a rheumatologist. Optimal management of AS requires a combination of both pharmacological and non-pharmacological treatment modalities.
Pharmacological
Non-steroidal anti-inflammatories (NSAIDs) medications are a first line treatment for AS and are proven to reduce pain, inflammation and disease activity. Management of dosage is done under the care of a rheumatologist. Long-term users of NSAIDs should be monitored for side effects, such as gastrointestinal and cardiovascular complications (12). Tumour necrosis factor inhibitors (TNF)-alpha inhibitors such as ustekinumab and tofacitinib are biological disease modifying anti-rheumatic agents (DMARD’s), which are proven to have large positive effects for AS (13).
Physiotherapy
The National Institute of Clinical Excellence (NICE) recommends that AS patients be referred to a physiotherapist for the commencement of an individualised exercise program, with periodic reviews to assess changing needs (6).
Benefits of exercise therapy
- Functional improvements
- Decrease systemic inflammation
- Decrease fatigue
- Reduction in co-morbidities (common in the AS population)
The European League against Rheumatism (EULAR) outlines that exercise programs should cover four major domains; aerobic, resistance, flexibility and neuro-motor exercise (11). High intensity exercise has historically been avoided in AS patients based on the theory that it exacerbates inflammation and therefore aggravates symptoms and pain. However, recent studies have concluded that this is not the case. A 2020 study compared the effects of a 3 month exercise programme with 3 months of treatment with a biologic immune-modulating TNFalpha-inhibitor. Results showed there was no significant difference between the two groups, with no reports of disease flare ups in either group (7). Not only was high intensity exercise safe, with no flare ups over the 3 month program, it was found to be as effective in disease modification as the TNFalpha-inhibitor.
The most important ingredient for any exercise program is patient adherence, which is generally quite poor (24-50%) in the AS population. A solid educational and reassurance base is required, particularly in disease populations, to explain the benefits of the exercise program and provide reassurance that adverse outcomes are extremely unlikely. Current research shows no significant difference in disease modification and patient functional outcomes between different modalities of independant exercise, only that exercise itself makes great improvements (11). Therefore, as with all physiotherapy management, exercise programs should be made in collaboration with patients, taking into account personal exercise preferences and disease progression. A significant difference is however noted between independent exercise programs and supervised programs, with greater adherence and improved patient outcomes when exercise programs are supervised by a physiotherapist or other qualified health professional.
Summary
Axial spondyloarthritis is a type of progressive inflammatory arthritis, affecting the lower back and sacroiliac joints. Management includes pharmacological and non-pharmacological techniques under the care of a Rheumatologist. Physiotherapy management for AS should include a regular home exercise program focussing on aerobic, resistance, flexibility and neuro-motor exercises. This can be combined with physiotherapist supervised exercise sessions for added benefits.

Eliana Lonsdale (APAM)
POGO Physiotherapist
Book an appointment with Eliana today
Featured in the Top 50 Physical Therapy Blog
References
- Magrey, M. N., Danve, A. S., Ermann, J., & Walsh, J. A. (2020). Recognizing Axial Spondyloarthritis: A Guide for Primary Care. Mayo Clinic Proceedings, 95(11), 2499-2508. doi:https://doi.org/10.1016/j.mayocp.2020.02.007
- Rehan, K., (2019). Ankylosing Spondylitis and Spine Surgery. Retrieved from: https://www.spineuniverse.com/conditions/spinal-arthritis/ankylosing-spondylitis/ankylosing-spondylitis-spine-surgery
- Hermann, K.-G. A., Althoff, C. E., Schneider, U., Zühlsdorf, S., Lembcke, A., Hamm, B., & Bollow, M. (2005). Spinal Changes in Patients with Spondyloarthritis: Comparison of MR Imaging and Radiographic Appearances. RadioGraphics, 25(3), 559-569. doi:10.1148/rg.253045117
- Wang, R., & Ward, M. M. (2018). Epidemiology of axial spondyloarthritis: an update. Current opinion in rheumatology, 30(2), 137–143. https://doi.org/10.1097/BOR.0000000000000475
- Brown, M.A., Kennedy, L.G., Macgregor, A.J., Darke, C., Duncan, E., Shatford, J.L., Taylor, A., Calin, A. and Wordsworth, P. (1997), Susceptibility to ankylosing spondylitis in twins the role of genes, HLA, and the environment. Arthritis & Rheumatism, 40: 1823-1828. https://doi.org/10.1002/art.1780401015
- National Institute for Health and Care Excellence. (2017). Spondyloarthritis in over 16s: diagnosis and management. Retrieved from https://www.nice.org.uk/guidance/NG65/chapter/Recommendations#pharmacological-management-of-spondyloarthritis
- Sveaas, S. H., Bilberg, A., Berg, I. J., Provan, S. A., Rollefstad, S., Semb, A. G., . . . Dagfinrud, H. (2020). High intensity exercise for 3 months reduces disease activity in axial spondyloarthritis (axSpA): a multicentre randomised trial of 100 patients. British Journal of Sports Medicine, 54(5), 292. doi:10.1136/bjsports-2018-099943
- Taurog, J., Chhabra, A., Colbert, R. (2016). Ankylosing Spondylitis and Axial Spondyloarthritis. The New England Journal of Medicine. 374;26.
- Dubreuil, M., & Deodhar, A. A. (2017). Axial spondyloarthritis classification criteria: the debate continues. Current opinion in rheumatology, 29(4), 317–322. https://doi.org/10.1097/BOR.0000000000000402
- Reveille J. D. (2015). Biomarkers for diagnosis, monitoring of progression, and treatment responses in ankylosing spondylitis and axial spondyloarthritis. Clinical rheumatology, 34(6), 1009–1018. https://doi.org/10.1007/s10067-015-2949-3
- Martey, C., & Sengupta, R. (2020). Physical therapy in axial spondyloarthritis: guidelines, evidence and clinical practice. Current Opinion in Rheumatology, 32(4). Retrieved from https://journals.lww.com/co-rheumatology/Fulltext/2020/07000/Physical_therapy_in_axial_spondyloarthritis_.7.aspx
- Kroon, F. P. B., van der Burg, L. R. A., Ramiro, S., Landewé, R. B. M., Buchbinder, R., Falzon, L., & van der Heijde, D. (2015). Non‐steroidal anti‐inflammatory drugs (NSAIDs) for axial spondyloarthritis (ankylosing spondylitis and non‐radiographic axial spondyloarthritis). Cochrane Database of Systematic Reviews(7). doi:10.1002/14651858.CD010952.pub2
- Ciurea, A., Scherer, A., Exer, P., Bernhard, J., Dudler, J., Beyeler, B., . . . on behalf of the Rheumatologists of the Swiss Clinical Quality Management Program for Axial, S. (2013). Tumor Necrosis Factor α Inhibition in Radiographic and Nonradiographic Axial Spondyloarthritis: Results From a Large Observational Cohort. Arthritis & Rheumatism, 65(12), 3096-3106. doi:https://doi.org/10.1002/art.38140







